A common obstacle to a successful market launch is the transition of medical products from design to manufacture, with issues often leading to unnecessary costs and significant delays. Several tips and techniques can be implemented to smooth the transition from the product development stage to full-scale production, and ensure products are delivered to market on time and on budget.
We provide a summary of 5 tips that can help smooth this transition. One key take-away - it is critical to include suppliers (i.e. metal stamping companies that manufacture the medical stampings used in medical devices) throughout the entire planning and production cycle.
You can read the complete article, written by Frank Pawlowski and Bill St. Onge of KMC Systems, Inc. as it originally appeared in Medical Design Technology.
1. Start Early
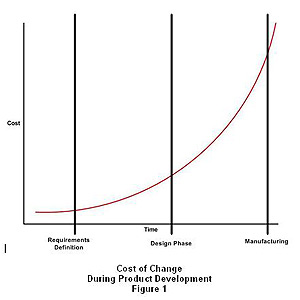
The first and perhaps most important tip to smooth the transition from design to manufacturing is to start early. Making changes to the product design once the handoff to manufacturing has occurred will only create additional costs and time delays. The “cost of change” increases asymptomatically as the product development cycle time increases.
Managing the “cost of change” also means making sure that the product requirements are solid before the design activities begin. This is paramount to reducing the risks associated with product changes during the development cycle. Further, making manufacturing a priority during the design process by involving production and quality control personnel as well as key vendors will help avoid additional design revisions at later stages. While change is inevitable, focusing the right resources early can minimize the probability of major change downstream in the development cycle or during manufacturing.
2. Assemble Cross-Functional Teams
Forming and utilizing cross-functional teams, comprised of mechanical, electrical, compliance, systems, manufacturing, and quality engineers, can also smooth the transition from design to manufacture. The team should include members of the supply chain, like buyers and planners, as well as suppliers, program managers, marketing personnel, and industrial design and human factor experts.
Cross-functional team members can work closely together to review product requirements, address technical risks, and manage instrument costs. Reducing development cycles to shorten time to market requires assembling the experts and cross-functional teams early to ensure manufacturability, serviceability, reliability, and a manageable supply chain.
The team environment is an important ingredient for a successful design to manufacturing product transition. Team members and vendors who are familiar with each other and have been through previous successful transitions can accelerate the process.
It should be noted that there are tradeoffs between moving through product development and transition to manufacturing too quickly, and bringing a superior product to market.
3. Rely on a Proven Process
Developing and continually tweaking the process used for transitioning products from design to manufacture will create a solid method for getting medical devices and instruments to market on time and within budget.
First, for a smooth handoff to manufacturing, planning for transition must begin in the product development stage well before the software and system verification and validation efforts have been completed.
A manufacturing test plan and detailed test specifications should be developed to include system level testing, sub-assembly testing, and spare part testing.
Once the test requirements are complete, specific test equipment should be designed and tested using the prototype system. The instrument’s on-board test capabilities can be utilized by end users in the field, eliminating the need for custom field test equipment.
During the pilot-build phase, the manufacturing team should begin developing detailed instructions for instrument assembly and setting up the production area. This will ensure rapid start-up once the transition phase is complete.
4. Implement Staged Reviews
Continual checkpoints should be held throughout the product design and development process, including critical design, pilot production readiness and manufacturing readiness reviews. A critical design review should occur at the completion of the product prototype phase and prior to the release of documentation for the procurement of pilot material. This review must assess product design to ensure that the hardware and software meet system requirements prior to pilot production.
The readiness of a product design to begin transition to manufacturing can be determined with a pilot production readiness review. The review will ensure that the system design meets all applicable specifications and customer requirements, and is mature enough for pilot production units to be built by operations.
A final Manufacturing Readiness Review (MRR) should be conducted after pilot production units are built and design validation is complete, to verify that the product is ready for full-scale manufacturing. The review will verify that all appropriate design, validation, and manufacturing preparations are complete and the program is adequately prepared to move to the production phase.
5. Train the Manufacturing Team
For a seamless and speedy manufacturing ramp-up, begin the training and knowledge transfer to the production team during product development and provide detailed assembly instructions. An effective training path will minimize wasted time and faulty products. Along with measures that increase and maintain morale and team spirit, the manufacturing team needs to be in tune with current Good Manufacturing Practices (cGMPs) as well as other FDA guidance regarding medical device manufacturing.
Conclusion
Whether an OEM plans to outsource manufacturing or not, the transition from design to manufacturing can either be a path to success or a roadblock to final product launch. Getting products to market on-time and within budget is highly dependent on the transition stage. Manufacturers that take manufacturability into account early and implement a proven process with cross-functional teams will reap rewards of condensed development cycles and larger profit margins.